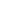Root Canal Irrigation with Dental Irrigants: A Review of EDTA and Similar Irrigants
2025-03-04 14:19:40
Introduction
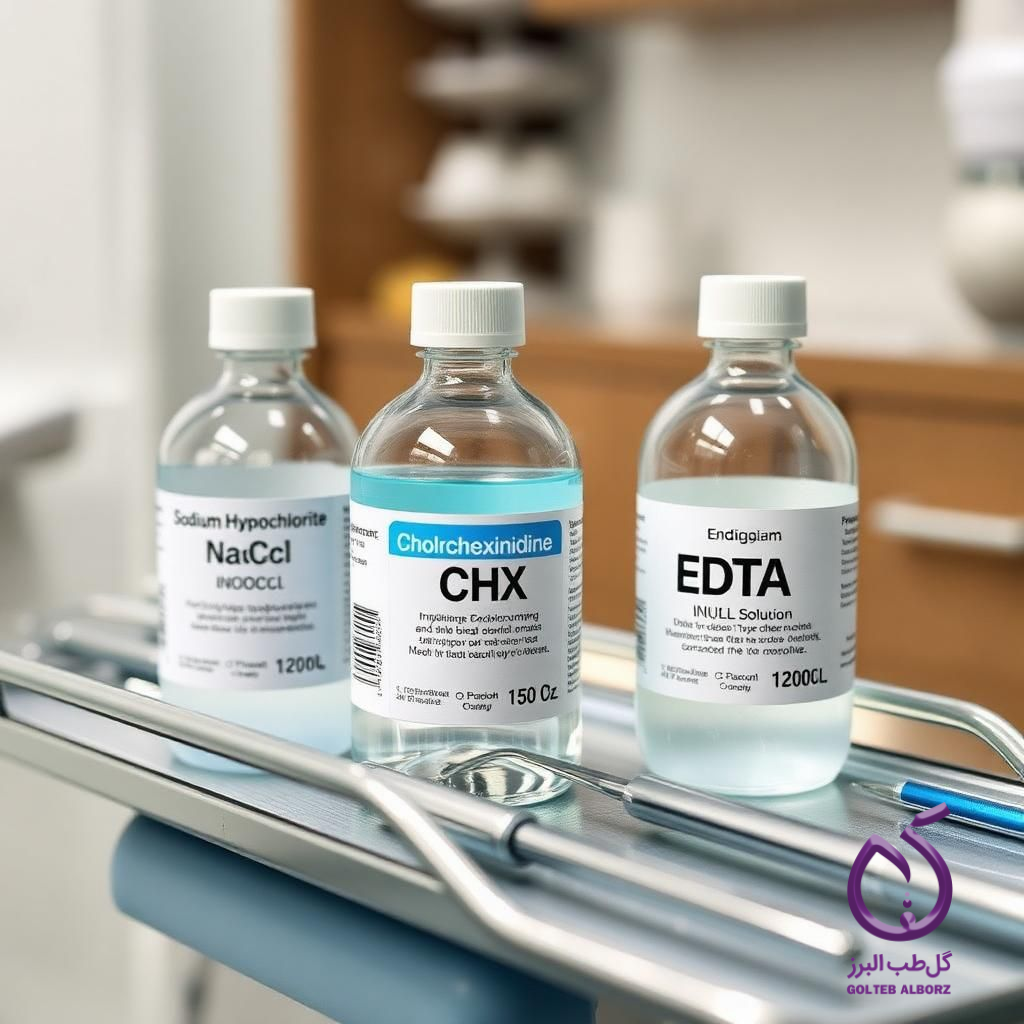
Root canal irrigation is one of the essential steps in endodontic treatment, significantly influencing treatment success. Various dental irrigants are used to remove both organic and mineralized debris from the root canal. Among these irrigants are ethylenediaminetetraacetic acid (EDTA), sodium hypochlorite (NaOCl), chlorhexidine (CHX), and salicylates. This article reviews the main irrigants used in dentistry and analyzes the advantages and disadvantages of each.
EDTA: Role and Applications
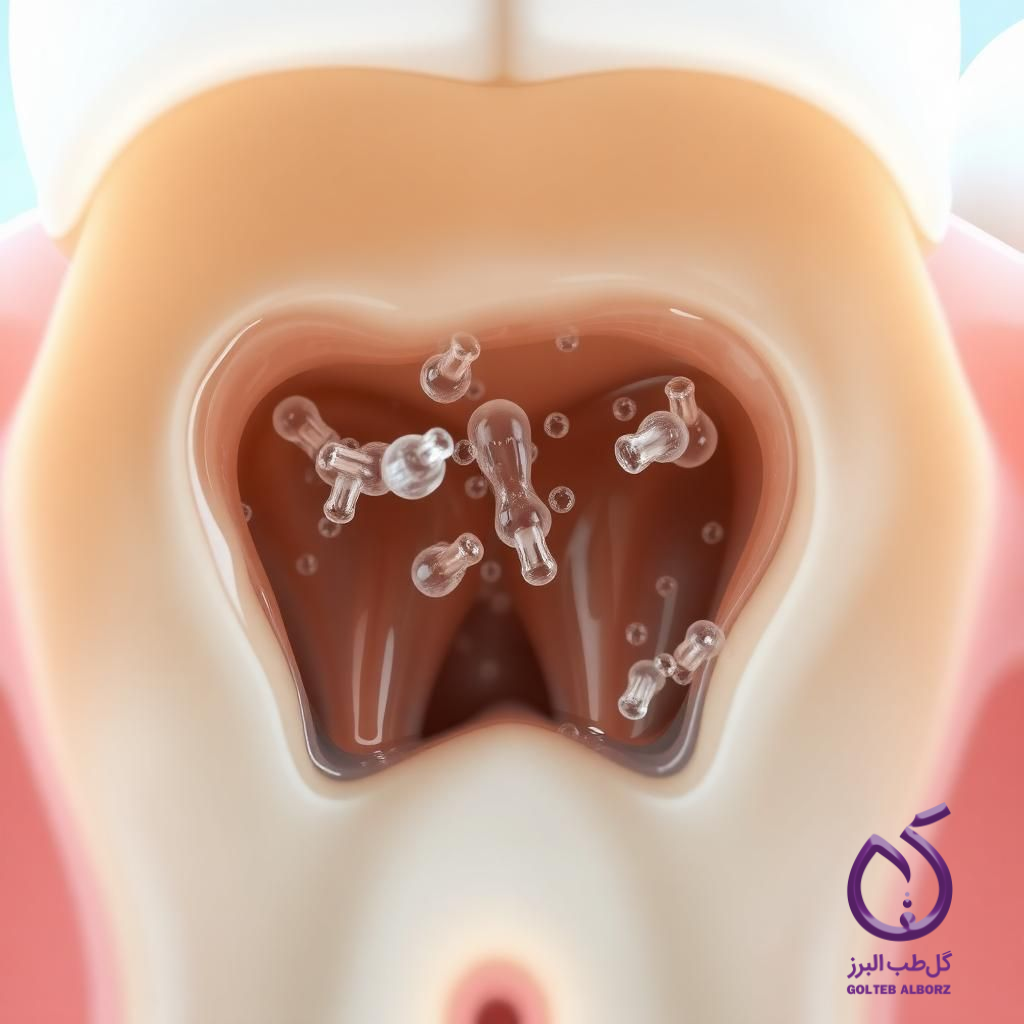
EDTA is a chelating agent used in dentistry to remove the smear layer and open dentinal tubules. By binding to calcium ions, it dissolves mineralized substances and better prepares the tooth surface for filling.
Chemical Properties and Function
EDTA is a weak acid compound used in concentrations of 15 to 17% for root canal irrigation. By removing mineralized debris from the smear layer, it cleans the canal walls and enhances the adhesion of filling materials to the dentin. EDTA is typically used alongside sodium hypochlorite to combine the dissolution of organic and mineralized components.
Advantages
- Effectively removes the smear layer.
- Reduces dentin resistance and improves penetration of filling materials.
- Increases the contact between disinfectants and canal walls.
- Opens dentinal tubules, enhancing the effectiveness of filling materials.
Disadvantages
- Unable to remove organic debris.
- Limited effectiveness in eliminating bacteria.
- Prolonged use may weaken tooth structure.
- Requires combination with other irrigants for optimal performance.
Sodium Hypochlorite (NaOCl)
Sodium hypochlorite is one of the most widely used dental irrigants, valued for its high tissue-dissolving capacity for necrotic tissue and strong disinfecting properties.
Chemical Properties and Function
Sodium hypochlorite is an alkaline chlorinated compound used in concentrations ranging from 0.5% to 5.25% for root canal irrigation. It can dissolve organic tissues and cleanse the canal walls of microorganisms. Its antibacterial properties, particularly against resistant bacteria such as Enterococcus faecalis, are highly effective.
Advantages
- Strong disinfecting properties.
- Ability to dissolve necrotic tissue and organic debris.
- Reduces the microbial load in the root canal.
- Capable of penetrating lateral canals.
Disadvantages
- Ineffective at removing mineralized substances and the smear layer.
- May cause sensitivity and irritation to surrounding soft tissues if extruded.
- Possesses a strong, irritating odor.
- Can react with chlorhexidine, forming a precipitate.
Chlorhexidine (CHX)
Chlorhexidine is a broad-spectrum cationic disinfectant widely used in dentistry for its long-lasting antibacterial effects and compatibility with oral tissues.
Chemical Properties and Function
Chlorhexidine is typically used at concentrations of 0.2% to 2% for root canal irrigation. It adheres to the canal walls, providing a prolonged antimicrobial effect. Unlike sodium hypochlorite, chlorhexidine does not dissolve necrotic tissue; however, its sustained action makes it an effective adjunct for reducing the risk of reinfection.
Advantages
- Strong antibacterial activity, particularly against Gram-positive bacteria and fungi.
- Provides a long-lasting antimicrobial effect within the canal.
- Does not irritate surrounding soft tissues.
- Effective against Enterococcus faecalis, a key contributor to recurrent root canal infections.
Disadvantages
- Unable to dissolve necrotic tissue and organic debris.
- May form precipitates and reduce effectiveness if combined with sodium hypochlorite.
- Prolonged use can potentially lead to tooth discoloration.
Comparison and Combination of Irrigants
In clinical practice, combining different irrigants is common to maximize their benefits. For example:
- The combination of EDTA and sodium hypochlorite allows for the simultaneous removal of organic and mineralized debris, thoroughly cleansing the canal.
- Chlorhexidine is often used as a final irrigant to provide sustained antimicrobial effects, although it should not be combined directly with sodium hypochlorite due to the risk of precipitate formation.
Latest Articles
Understanding MID: How It Relates to Pulpotomy and Pulp Capping
Read more
Speed Up Tooth Socket Healing with Ora-Aid Oral Dressings
Read more
Bioceramic Materials in Pediatric Dentistry
Read more
Ora-Aid oral adhesive – overview & benefits video
Read more